What leads to more spike protein in the body: the vaccine or virus? Part 2
A mini-review of what we know
Brief Background
The SARS-CoV-2 virus has a protein on its surface called the spike protein. The COVID vaccines available in the U.S. work by getting the body to produce this protein, with some modifications.
In a previous article, I went over multiple pieces of evidence showing that the spike protein alone, either from the virus or vaccine, was harmful.
In Part 1 of this article, I responded to an article by Uri Manor and Jeremy Howard. Their article claimed that:
(1) The amount of spike protein from the COVID vaccines was harmless or physiologically negligible.
(2) The amount of spike protein from the COVID vaccines was much lower than what one would get under viral infection from SARS-CoV-2.
They used these claims to argued that if you were trying to gain immunity, it was safer to get it via the vaccines than with infection by the virus.
In Part 1, I argued that the study that Manor and Howard used to make claim (1) did not actually measure all physiologically relevant spike protein. I also argued that we already had evidence that the levels of spike protein from the vaccines were physiologically relevant.
In this article, I’ll examine claim (2).
The claim
In their article, Manor and Howard say that the levels of spike (S) protein found in vaccine recipients are extremely small in comparison to what one would get under viral infection.
They cite two studies to back up their claims: one by Sender et al., which they use to estimate the number of virus particles one would be exposed to under infection, and another by Neuman et al., which they use to estimate the number of spike proteins on each virus particle.
From their article:
…humans may have what some have estimated to be as high as 1 to 100 billion virions in the lungs [Sender et al., 2020]. Given there are approximately 100 spike proteins per virion [Neuman et al., 2011], this means COVID-19 infections could in theory result in as many as 10 trillion spike proteins.
When Manor and Howard say “Given there are approximately 100 spike proteins per virion [Neuman et al., 2011],” I can only assume they are referencing this part in the Neuman et al. paper:
To test the quality of this model, we counted the number of spikes on three-dimensional reconstructions of MHV particles. These particles showed an average of 74 spikes per particle, which gives an approximate inter-spike spacing of 17 nm. Our model predicts ∼90 spikes per particle.
So their “100 spike proteins per virion” number seems to be based on a model for MHV (a coronavirus of the mouse).
Now in the absence of any real data, it makes sense to use a model of another coronavirus to make estimations of the number of spike proteins on SARS-CoV-2. But we now have a more recent study which was published in Nature, that used cryo-electron microscopy to count the number of spike (S) proteins on SARS-CoV-2, and here’s what they found:
Individual virions contained 24 ± 9 S trimers (Extended Data Fig. 1b). This is fewer than previous estimates that assumed a uniform distribution of S, because S was not uniformly distributed over the virus surface.
So that’s quite a bit lower then the estimate that Manor and Howard used.
As far as the Sender et al. study goes; this paper took studies that looked at tissue concentrations of virions in rhesus macaques and extrapolated the number of virions in humans.
According to that estimate, there would be 1 to 100 billion virions in the lungs under infection.
From here, Manor and Howard argue that this leads to a lot more spike protein under infection, compared to vaccination.
Examining assumptions
Manor and Howard seem to assume that these estimates are sufficient to decide that vaccination is a safer way to gain immunity, compared to infection.
But this is incomplete, and lacks nuance.
First of all, we know that vaccinated people can still get infected; the vaccines do not prevent one from getting infected.
With the possible exception of hermits, everyone is going to get exposed to this virus at some point.
The question then becomes whether vaccination lowers the amount of virus produced under infection, and if so, to what degree. That would allow us to know whether the difference in viral load under vaccination is “worth” the extra spike protein one gets exposed to from the vaccine.
Now this gets tricky, given that we don’t know how much spike protein gets produced as a result of vaccination, and we don’t know to what degree it varies across individuals. Unfortunately, this was not studied.
It should also be made clear that the amount of spike protein is not the only relevant factor in safety; it also matters how it is delivered (all at once, or gradually, within a viral capsid, or within LNPs), etc.
But let’s examine what we do know.
What do we know about how vaccination affects the amount of virus (or free spike protein) one gets exposed to?
In an ideal world, we would be able to measure the amount of all physiological relevant spike protein, whether it was free spike protein or spike protein attached to virus, and account for all of it in the body during the course of an infection, and compare the levels between the vaccinated and unvaccinated.
If we wanted to do that we would have to look at the amount of virus or spike protein in:
In the upper respiratory tract
In the lower respiratory tract, including lungs
In plasma
In other places, such as within the lymphatic system, on cells, within cells, or on exosomes
Let’s look at what data is out there for each of these.
Examining assumptions 1- Virus in the upper respiratory tract
Various studies here and here and here, took nasopharyngeal swabs from fully vaccinated and unvaccinated individuals and used PCR threshold cycle (Ct) values as proxies for Delta variant viral load.
In each of these cases, they found that there was no significant difference in viral load between the two groups.
On the other hand, a study from Singapore, found that viral loads were similar between the vaccinated and unvaccinated initially, but decreased faster in vaccinated people (see Figure 1).
None of these studies were perfect; for example in the Singapore study, the median age in the vaccinated group was higher than in the unvaccinated, but on the other hand a much larger percentage of the unvaccinated were male or had diabetes mellitus (Table 1).
Also, what was excluded from these studies was any measure of viral load from individuals who were only partially vaccinated. “Fully vaccinated” was defined as those who’d had their final dose for at least two weeks. These measurements would have been interesting to have, because there is data that indicates that there is a window of time post vaccination, where one is somewhat immunocompromised and more susceptible to infections for about two weeks (see Fig 2 of this paper).
[UPDATE 12/15/21- A caveat: that study looked at elderly people who had been vaccinated. We don’t know whether we’d see that same effect in younger people].
[UPDATE 12/18/21- On the other hand, see Extended Data Fig 1 in this study, which shows lower lymphocyte counts initially after the Pfizer vaccine.]
But for now let’s say it’s possible that those who have just gotten their first dose and are unlucky enough to have gotten infected within that window of time, would have a worse infection (and produce more virus) than they would have otherwise. We don’t know for sure, however, since there doesn’t seem to be good data on this.
It would require a lot more space to fully dissect these studies, but let’s grant that viral loads at peak levels seem to be similar in the vaccinated and unvaccinated, and vaccination may lead to lower viral load later in the course of infection.
Let’s also say that it’s unclear whether viral loads would be higher for those partially vaccinated individuals who happen to get infected within two weeks of their first dose.
Examining assumptions 2- Virus in the lower respiratory tract, including the lungs
As far as I know, there aren’t any studies that compare the amount of virus produced in the lower respiratory tracts of the vaccinated vs the unvaccinated, but evidence in animal models suggests that vaccinated animals have lower numbers of virus in the lower respiratory tract under infection, compared to unvaccinated animals.
For example, here was a study where an intramuscular DNA vaccine that encoded for the spike protein resulted in lower viral loads in the lungs in Syrian hamsters, which are highly susceptible to SARS-CoC-2 infection. However, it took 3 doses of the vaccine to really see this effect.
The graphs below show measurements of SgE (sub-genomic E, or envelope) copies which are proxies for viral load. IM means the vaccine was injected intramuscularly and IN means it was intranasal.
There wasn’t a huge difference between the treatments in the first graph, which compares prime + boost vaccinations with controls (empty or saline) but in the second graph, where the hamsters got prime + boost + boost, aka three intramuscular injections (IM-IM-IM), there was lower viral load in the lungs compared to the control (empty).
Somewhat surprisingly, the IM-IM-IM group tended to have lower viral load compared to the IM-IN-IN group. One would think that the hamsters that had some IN (intranasal) delivery would get less viral load in the lungs, but that’s not what we see.
Also surprising: with the exception of a single hamster in one of the vaccinated groups, lung pathology was similar in all groups. They also saw exacerbated pathology in the lungs of the hamsters that received the IM + IN vaccinations relative to the IM only or control hamsters. Although the authors of the study do not discuss this, I’m going to guess this is because the spike protein encoded by the vaccine is toxic, and administering it directly to the lungs will lead to some amount of lung damage.
In another study here, viral RNA was measured in the lungs and lower respiratory tract in rhesus macaques, with or without the ChAdOx1 (AstraZeneca) vaccine. When exposed to the virus, vaccinated animals had reduced viral load in the lungs and lower respiratory tract tissue compared to control animals.
Oddly, viral RNA in intestinal tissues like the duodenum, jejunum, etc. was higher in vaccinated animals (especially those that had a booster shot) compared to controls (see below).
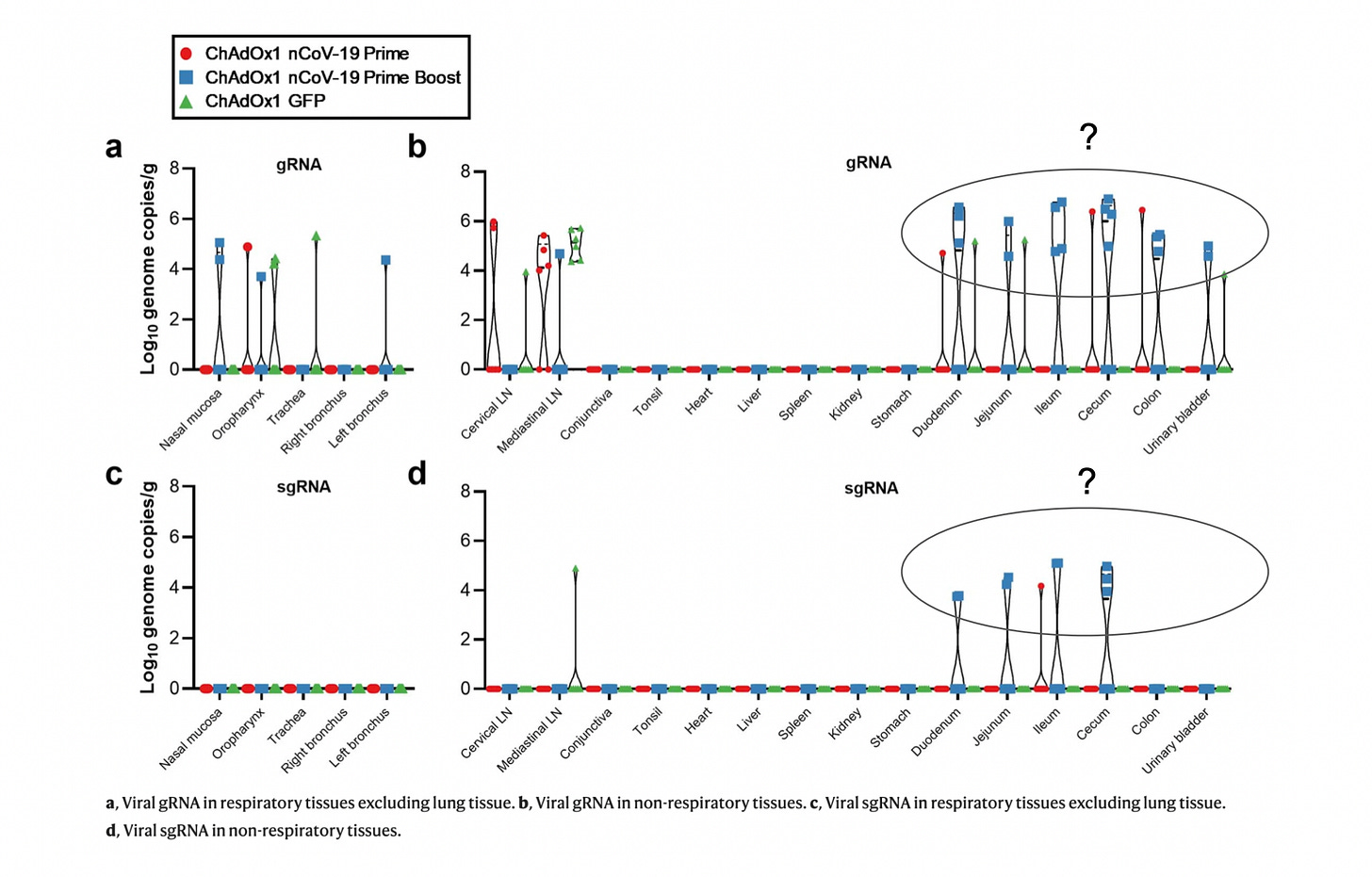
Something to keep in mind is that the animals in these studies were challenged with high levels of virus.
But based on this information, let’s grant that with the right dosing regimen, intramuscularly injected vaccines can lower viral load in the lungs.
Examining assumptions 3a- Virus in the plasma
Viremia is a condition where virus has leaked into the blood. An individual can have an active viral infection in the lungs, but this does not mean that virus will get into the blood.
How common is viremia with SARS-CoV-2 infection?
In this study, researchers quantified SARS-CoV-2 viral RNA (vRNA) by ultra-sensitive RT-PCR in plasma samples from 51 COVID-19 patients. The source of the vRNA in the plasma would presumably be from some breakdown in the barrier of the lungs during infection.
The patients fell into three groups: (i) ICU patients that were critically ill, (ii) non-ICU inpatients hospitalized patients, and (iii) outpatients, or those who visited the hospital but didn’t stay overnight.
What they found was that all 23 of the ICU patients, 10 out 19 non-ICU hospitalized inpatients, and 1 out of 9 outpatients, had detectable SARS-CoV-2 vRNA in their plasma. See below:
Using vRNA in the blood as a proxy for viremia, here are some thoughts:
Patients with more serious cases were more likely to have detectable vRNA in their plasma. This wasn't surprising.
I’m not aware of any studies looking into whether vaccination leads to less viremia after infection, but my guess would be that if vaccination leads to less virus in the lungs, and milder symptoms, it would lower the risk of viremia.
I’d also guess that if viremia does occur, vaccination would help contain the virus in the plasma to some degree, since neutralizing antibodies against spike protein should be present.
What was interesting was that just 1 out of the 9 outpatients had any detectable vRNA in their plasma, and that level of vRNA was extremely small (keep in mind that this study used an ultra-sensitive detection method).
Also keep in mind that although outpatients had mild illness compared to inpatients, they still had enough symptoms to warrant going to the hospital.
This should make us wonder whether viremia would be found in people who don’t go to the hospital when infected, either because they have even milder symptoms, or no symptoms at all.
That should also make us wonder to what degree vaccination would do anything to prevent viremia in young healthy people because they would be at such low risk of viremia anyway.
Examining assumptions 3b- Free spike protein in the plasma
This study measured free spike or spike S1 subunit in the plasma of infected individuals.
All these patients were admitted to the hospital, and they were categorized as “Floor admission” or “Floor admission then ICU later in hospitalization” or “ICU.”
In the graphs below are just two examples of patients that went to the ICU. The part to pay attention to are the graphs on the right, particularly the red circles which represent the S1 subunit.
What was surprising was that these patients had no detectable levels of S1. These were patients that went to the ICU. Patient 29 was 63 years old and patient 30 was 72 years old.
Now, there were certainly some patients that had measurable S1 levels, but roughly 40% had either none, or virtually none, and we’re talking about patients that have been hospitalized.
Here’s a subset of the patients, where the ones who had no detectable levels of free S1 are highlighted in yellow:
The yellow does not even include patients who had almost no S1.
Now it’s possible that we sometimes see zero detectable spike because of when the first plasma samples were taken; perhaps in some of these cases there was some free spike protein in the plasma near the beginning of the infection, and by the time the first plasma sample was collected, the level of spike had gone to zero.
However, I bring this up to make the point that we can’t assume that infection automatically leads to high amounts of free spike protein in the blood. What we see here is that even in hospitalized patients, we sometimes don’t have measurable levels of spike in plasma.
So my guess is that if we looked at healthy infected individuals that were not hospitalized, that almost none would have free spike in the plasma. That’s just a guess.
Examining assumptions 4- Virus or spike protein in other places
Some of the other places one could find either virions or spike protein include: in lymph, on cells, within cells, or on exosomes.
What we do know is that circulating exosomes with spike proteins have been found from infection and vaccination.
And the work of Dr. Bruce Patterson and his collaborators have shown that both infection and vaccination could sometimes lead to spike S1 subunit lingering within non-classical monocytes, which could lead to long COVID or COVID-like symptoms.
There isn’t much data quantifying and comparing these in the vaccinated vs the unvaccinated.
[UPDATE 2/14/22- A new paper that was published in the journal Cell, measured spike protein and vaccine mRNA in lymph nodes of people who were vaccinated with the BNT162b2 (Pfizer-BioNTech) vaccine. They found both spike protein and mRNA from the vaccine was still present 60 days post-vaccination. For mRNA to last this long is unheard of; “natural” mRNA is not known to last this long, but the mRNA of the vaccines is modified. One of the modifications that could be responsible for its longevity is the fact that N1-Methylpseudouridine was used to replace uridine. Also, 60 days was the maximum number of days that were studied so it’s entirely possible that the mRNA can last even longer.
Side note: The CDC website claims that the mRNA of the vaccines is broken down after a few days (see “Facts About COVID-19 mRNA Vaccines”). They said this even though they had no way of knowing this at the time.]
Summarizing what we think we know
Let’s summarize what we think we know and make some educated guesses, based on the limited data we have:
Vaccination doesn’t prevent infection, so vaccinated people are still going to be exposed to some virus.
There is a window of time shortly after vaccination when the recipient is somewhat immunocompromised, so it’s conceivable that if the person gets infected during this time, viral loads might be higher than they would have been without the vaccine.
Vaccination does not seem to significantly lower the amount of virus in the upper respiratory tract, based on nasopharyngeal swabs.
Viral load might decrease faster in the upper respiratory tract of vaccinated individuals, according to the Singapore study.
Vaccination might lower the amount of virus in the lower respiratory tract, based on animal studies.
Vaccination could conceivably lead to more virus in other tissues, as was found in the rhesus macaque study.
On the other hand it seems conceivable that infection without vaccination could lead to more virus in other tissues (this is just conjecture on my part, as I want to “steel man” the vaccination case).
If vaccination lowers the amount of virus in the lungs, it would make sense that it might help prevent viremia, though I’m not aware of studies showing this.
Vaccination would theoretically help prevent the virus from replicating if viremia does occur, but since viremia is somewhat rare in the first place, this might only help patients that have more severe cases.
Free spike protein in the plasma is not found in even a lot of hospitalized patients, so it is probably found even less in mild cases.
We don’t have good data comparing viral or spike protein levels in the vaccinated vs unvaccinated in other reservoirs, like in or on cells, on exosomes, etc.
The “efficacy” of the vaccines (and by “efficacy” most people are talking about antibody levels) wanes after some number of months. Given this, if vaccination does lower viral levels under infection for several months, the best course of action might be for the vaccinated person to try and get infected before their vaccine efficacy wanes. After that, they would have natural immunity, which seems to be longer lasting, and better at staving off reinfections (see here, here, and here).
Each time someone gets a dose of the vaccine, they are subjecting themselves to unknown amounts of spike protein. Damage from the spike protein, or other components of the vaccine, might increase in a dose dependent matter. This damage could be subclinical.
Under natural infection, natural barriers in the nose and mouth mean that in young healthy people, there could be a good chance that spike or the virus won’t get into places beyond the lungs. With vaccination spike (or the mRNA encoding the spike) could end up getting to places where it otherwise might not have reached.
One probably gets more spike protein from each unit of vaccine mRNA, compared to a unit of viral RNA. This is because the mRNA of the vaccines was codon optimized and altered (N1-Methylpseudouridine was used to replace uridine) so that it lasts longer and is translated more efficiently, compared to “natural” RNA.
[UPDATE 12/15/21- People who have been vaccinated have higher peak levels of anti-spike antibodies levels, compared to people who have only been infected. This suggests that the vaccinated are exposed to higher levels of spike protein, though we can’t say this for certain.]
This summary is what I arrive at after piecing together data from several different sources. I don’t expect to be right on all of these, and expect that I’ll update my views as we get more data.
The main point is: trying to determine whether someone who is vaccinated will ultimately be exposed to less spike protein, compared to an unvaccinated person, is not a simple thing to do.
A missing piece of the discussion: steelmanning the infection case
What was odd about Manor and Howard’s article was that the only alternative to infection that they considered, was vaccination. They didn’t consider the case of infection with simple health interventions like vitamin and mineral supplementation, which could tip the scales in favor of infection.
We know that maintaining adequate vitamin D (see here and here), C, and zinc levels play a huge role in COVID outcomes.
What if we steel manned the infection case, and measured viral loads in patients who were infected but had adequate levels of vitamin D, C, and zinc?
Manor and Howard also didn’t consider whether the risk-benefit ratio made sense for younger people.
We know that younger people, especially children, have very low odds of adverse clinical outcomes from COVID-19.
What would all the measures of viral load, in either the upper respiratory tract or the lungs or in the blood, look like if we only focused on younger people?
On top of that, what if patients could be given drugs or early treatments that some front line doctors have vehemently advocated for; treatments that act as antivirals, anti-inflammatories, or both?
Does it seem clear from all this that gaining immunity through vaccination is always better than getting it through infection?
Immunity in younger people
We can’t assume that children would produce remotely the same amount of virus under infection compared to adults. There are major differences between the immune systems of children and older people, and this warrants further discussion.
Immunity can be categorized into two broad categories: innate immunity, which does not require prior exposure to an antigen (like an invader), and acquired immunity, which does require prior exposure. Although the vaccines can stimulate components of innate immunity, the main mode of action of the current COVID vaccines seems to be via acquired immunity, by getting one’s body to produce anti-spike antibodies; this is based on the fact that their efficacy seems to wane with anti-spike antibody levels.
In this study, researchers found that children who had SARS-CoV-2 infection had lower and reduced breadth of anti-SARS-CoV-2-specific antibodies compared to adults, and had reduced neutralizing activity compared to adults. Yet we know that children with SARS-CoV-2 infections have milder cases with significantly less morbidity and mortality compared to adults. This suggests that in children, a more robust innate response in the course of infection is perhaps “chosen” over a vigorous adaptive response.
Some concrete evidence for this was in a recent study in Nature, where researchers found large differences in the composition of the immune or epithelial cells (which line the mucus membranes) in the nasal mucosa. Among other things, they found that children had much higher levels of neutrophils, which are a type of phagocytic cell, and part of the innate immune response. They also found that some epithelial cell populations showed age dependency; for example goblet cells, which play a role in mucus secretion, decreased, and ciliated cells increased, with age.
Additionally, the genes for pattern recognition receptors (PRRs), which are receptors that are supposed to recognize foreign invaders like microbes or viruses, were expressed at much higher levels in the epithelial cells of the upper respiratory tract in healthy children, compared to adults. This suggests an increased ability of the respiratory mucosa of children to quickly respond to viral infections.
Much more could be said on this topic, including the natural mucosal barriers in children, but the main point is this:
We can’t assume that the risks of the vaccines are worth the benefits, especially when it comes to children.
This is not quite the end
In their article, Manor and Howard make a lot of assumptions about the level of virus one would be exposed to under infection, and leave out subtleties like age or simple things that could be done to improve outcomes under infection, like having good vitamin D status.
They also don’t consider all the other factors involved in vaccine safety, like the potential risks associated with delivery mechanisms like LNPs (lipid nanoparticles), or the fact that uridine was replaced with a pseudouridine in the mRNA vaccines, or the liberal use of codon optimization (see a recent paper on this here).
I suspect that part of the reason that many in the extremely pro-vaccine camp don’t put as much weight on the potential risks of the vaccines, is because they assume that adverse events from the vaccines are extraordinarily rare.
I think there is now plenty of evidence that adverse events from these vaccines are grossly underreported.
More on that in a future post. You can subscribe or follow here for updates.









